
Aluminium is ubiquitous in the environment. In the last few decades, it has increasingly been incorporated in consumer products, food and drinking water with a presumption of safety. This presumption is based on the belief that as is not overtly toxic in acute low dose ingestion, cumulative consumption must also be benign. Excess aluminum can contribute to a deterioration of mental health. Excess aluminium has also been implicated in the development of Alzheimer’s disease.
Aluminium in Water
To date, no physiological requirement for aluminium in human health has been found. (1) Contrary to the popular assumption of safety, chronic low-level ingestion exceeding 1 g per day has been implicated in several conditions including hypophosphataemia (caused by aluminium forming insoluble compounds with phosphate), elevated calcium level, bone resorption (building), osteomalacia (softening of the bones) and Alzheimer’s disease. Embryotoxic effects have also been shown in preclinical trials.(1,2) Along with the total limit for ingestion, a ‘safe’ drinking water limit of 0.6 mg/ L or 600 ppm per day has been proposed, based on a 40-day aluminium balance study.(1)

Aluminium is used in cookware, as takeaway containers and as a food foil for baking and food storage. Research has confirmed that it is more likely to leach into acidic foods such as lemons and oranges and some salty protein rich foods such as salmon and ham. (2) Yet, how often we see fish wrapped in aluminium foils and being barbequed or cooked. Did you know that leaching effect increases up to 18 fold when food is cooked and/or stored in aluminium foil on a tray made of an alloy with a higher electrical potential than aluminium, for example, stainless steel. (2) Aluminum is also added to common salt and icing sugar so it does not absorb humidity. Therefore, it is advised to use Celtic or Himalayan salts and make your own icing sugar.
Aluminium in Personal care

However, the most common route of aluminium exposure is through the use of antiperspirants which contribute approximately 2 g of aluminium per day to the skin. This is absorbable, not directly via the skin epidermis but rather via the hair follicles, apocrine and eccrine sweat ducts.(1) Additionally, certain sunscreen can contribute up to 5 g per day.(1) Aluminium is also commonly found in consumer and personal care products; pharmaceutical products such as antacids, adjuvants and prescription medications and is used to remove sediment from drinking water. The residual aluminium left in the water can range from 1.17 mg/L to 5.35 mg/L.(3)
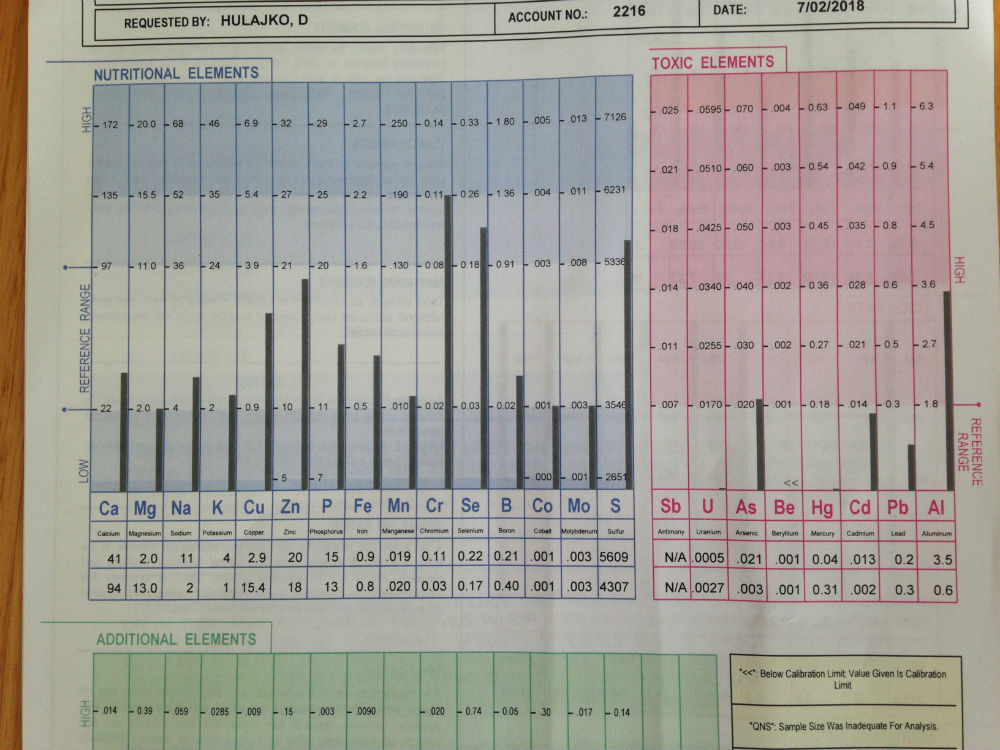 Recently I saw an 82 years old patient with a very high level of aluminum, (image above) which was not shown in her previous four mineral hair analyses done over a period of ten years. Reason being that her elevated copper and lead were chelated first. This patient was using aluminum pots and aluminium foil when she was young. This means that all those years aluminium was accumulated in the patient’s body, waiting to be chelated. Also, it is evident that the patient is malnourished due to her poor absorption and her age. Iron, zinc, calcium and magnesium antagonise aluminium and those minerals are low in the patient. Supplemention with those minerals as well as with vitamin E and C will ensure chelation of aluminium.
Recently I saw an 82 years old patient with a very high level of aluminum, (image above) which was not shown in her previous four mineral hair analyses done over a period of ten years. Reason being that her elevated copper and lead were chelated first. This patient was using aluminum pots and aluminium foil when she was young. This means that all those years aluminium was accumulated in the patient’s body, waiting to be chelated. Also, it is evident that the patient is malnourished due to her poor absorption and her age. Iron, zinc, calcium and magnesium antagonise aluminium and those minerals are low in the patient. Supplemention with those minerals as well as with vitamin E and C will ensure chelation of aluminium.If you would like to know more about how the DH-Natural Medicine Clinic can help you, please call us now on (02) 4854 0205
 Danuta Hulajko is a holistic practitioner, international speaker and the founder & practitioner at the DH Natural Medicine Clinic and www.healingremedies.com.au in the Southern Highlands.
Danuta Hulajko is a holistic practitioner, international speaker and the founder & practitioner at the DH Natural Medicine Clinic and www.healingremedies.com.au in the Southern Highlands.
Danuta specialises in Allergies, Anti-Aging, Auto-Immune Conditions, Cardiovascular Conditions, Female Reproductive, Menopause, Mould Toxicity, Skin Conditions, Stress and Insomnia and Thyroid Dysfunction.
For more information please go to our website. You can also follow Danuta Hulajko’s work, events, seminars, expos, latest health research, her health tips and advice on Facebook, LinkedIn and Instagram.
- Exley C. Human exposure to aluminium. Environmental Science: Processes & Impacts. 2013;15(10):1807-16.
- Ertl K, Goessler W. Aluminium in foodstuff and the influence of aluminium foil used for food preparation or short time storage. Food Additives & Contaminants: Part B. 2018 Feb 19.
- Alexeeff, George V. Public health goal for aluminium In drinking water. 2001

